UV Beads Experiment & The Electromagnetic Spectrum
This UV beads experiment is a super-popular activity with kids. To help students understand why the beads change color, they need to understand ultraviolet light.
While studying the electromagnetic spectrum, you may want to add a study of solar energy or this sun print activity–it dovetails nicely with the uv bead experiment in this post.
I want to add a personal note, as I think this activity is more than just watching the beads change and the “cool” factor that has. My mother passed from melanoma. It was a very aggressive and, in the end, painful form of cancer. My children were old enough to understand and to this day, are very careful in the sun. I hope that this activity will also teach students that UV light is present, even on cloudy days, and it’s important to protect our skin.
UV Beads Experiment and The Electromagnetic Spectrum
Microwaves, cell phones, and RADAR are modern-day inventions that utilize the electromagnetic spectrum, but the fundamental groundwork for the study of electromagnetic waves started with the ancient Greeks, who first discovered static electricity. In the 11th century, the Chinese described magnetic properties.
However, it wasn’t.t until the 1800s when James Clerk Maxwell claimed electricity and magnetism were related.
He established some basic facts and equations to prove his theories. He believed:
- An electrical charge creates a field
- Magnets have two poles, North and South
- A magnetic field creates an electric current
- An electric current creates a magnetic field.
In addition, Maxwell said waves of energy existed and traveled at the speed of light (Scientists had started to study this as well). He claimed that visible light wasn’t the only form of electromagnetic energy.
Many people thought Maxwell and his ideas were wrong, but scientists proved Maxwell was correct.
One such scientist was Henry Hertz. In 1888 he discovered radio waves. Unfortunately, he died at the young age of 36.
Other scientists and inventors, such as Nikola Tesla and Guglielmo Marconi, used Hertz’s discovery to patent many inventions.
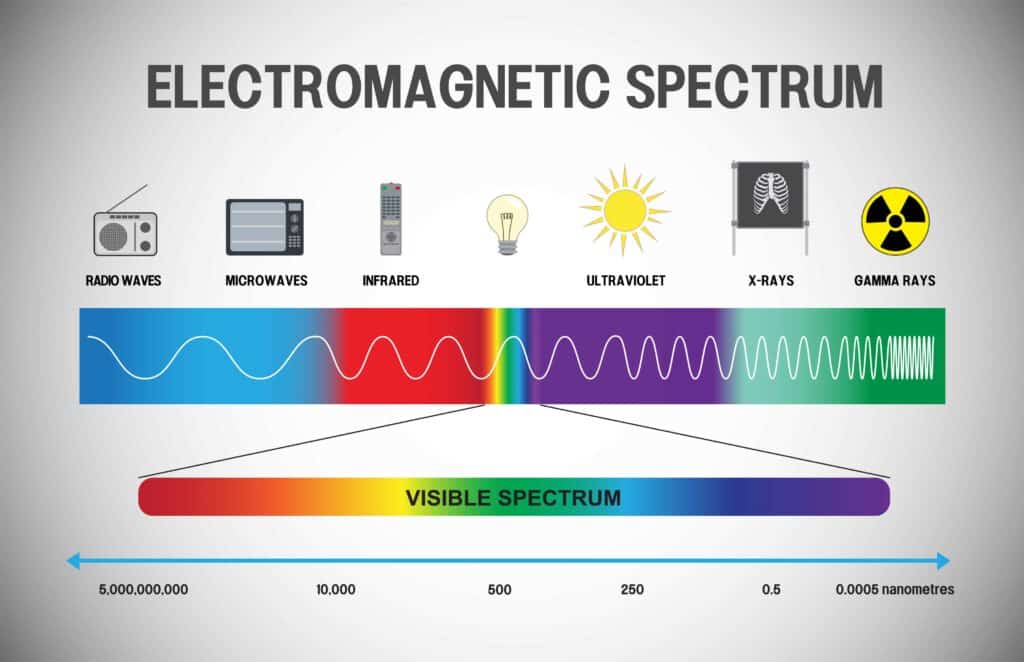
What is the Electromagnetic Spectrum?
Do you use a microwave, radio, cell phone, or television? These objects use at least one form of electromagnetic wave. Man-made objects may use electromagnetic waves to function, but the Sun and other objects in the universe emit electromagnetic radiation. To understand the electromagnetic spectrum, we will first define electromagnetic radiation. Let’s start with the term electromagnetic. Electromagnetic is another word for light.
Light is fluctuations in the electric and magnetic fields. Stars, planets, and the Sun all have magnetic fields. In fact, all objects with a temperature above absolute zero radiate electromagnetic radiation. Even ice cubes emit thermal radiation!
Electromagnetic radiation is energy emitted in the form of particle waves. Unlike sound waves, electromagnetic waves do not need air or water in which to move. In fact, electromagnetic waves are the only form of waves that can travel through empty space, such as the vacuum of the universe. In addition, all electromagnetic waves move at the speed of light. How fast does light travel?
The speed of light is approximately 299,792,458 meters per second (or about 186,282 miles per second). This is a fundamental constant of nature and is the maximum speed at which any form of energy or information can travel through space.
Scientists can determine what stars are made of by using a spectroscope to analyze the electromagnetic radiation emitted by the star.
Electromagnetic radiation is energy traveling in the form of particle waves and carrying a certain amount of energy. This amount of energy varies.
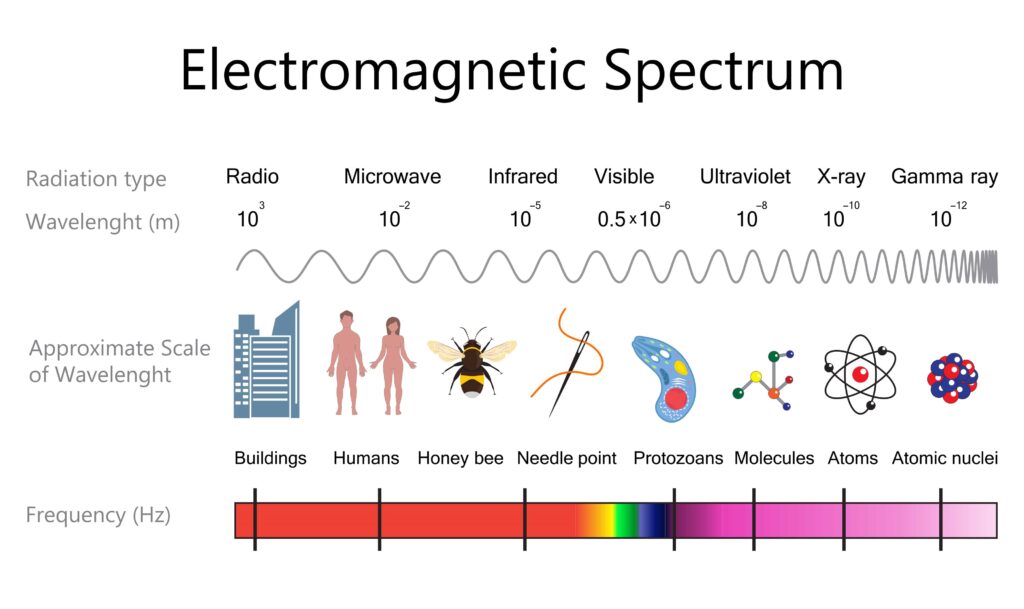
Radio waves are the weakest, and gamma rays are the strongest. Energy is directly related to temperature. Longer waves have a lower frequency and contain less energy. Shorter waves have a higher frequency and contain more energy.
The size of the waves in the electromagnetic spectrum range anywhere from the size of mountains to the size of atoms, and even smaller. Radio waves are big, and the antenna on your radio is as big as the radio waves it receives. Microwaves are small enough to fit and be useful in a microwave oven.
Infrared can be detected in something as small as the scope of a rifle. Radio and microwaves won’t hurt you. Infrared you can feel as heat. Ultraviolet-A and UV-B can burn you easily and cause enough cell damage in your skin that mutations from the damage can lead to cancer. X-rays are given to you in small enough doses by a radiologist (For example, if you break a bone and need X-rays.) that you are not injured. But think about it; the x-rays pass right through your body and expose the film on the other side of you, and they cover you with a lead shield to keep your exposure to a minimum. Gamma rays are what a Geiger counter detects and are what we think of as nuclear radiation.
Night vision goggles can detect a human being’s body in complete darkness. We think of it as complete darkness because we cannot see any visible light. However, our bodies are infrared radiators, given our body temperature of 98.6°F (On average. Some people will register a slightly lower body temperature while others register a higher temperature.). In most parts of the world, the ambient temperature is less than 98.6°F, so, our bodies are brighter sources of infrared radiation than the environment and stand out like light bulbs. Infrared radiation is an indicator of a relatively low level of energy or heat.
Scientists use radio telescopes to detect X-ray emissions throughout our universe. Sources of X-ray radiation are extremely hot. The events that cause the release of X-radiation are usually violent and unimaginably powerful. X-rays indicate high levels of energy or heat.
Visible Light Spectrum
Visible light is part of the electromagnetic spectrum that we can see using our unaided eyes.
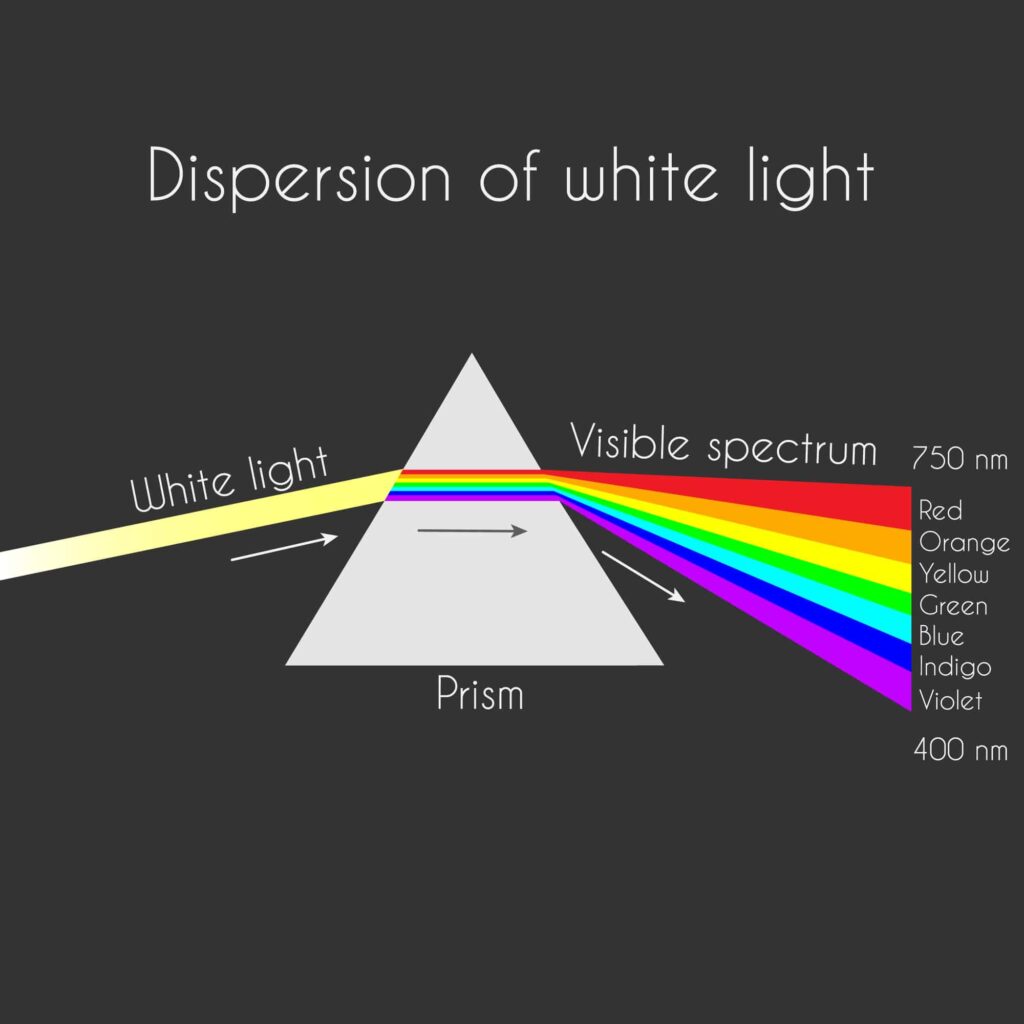
Sometimes you might hear the term white light. The light has color, but we cannot see the colors until we view a rainbow in the sky or direct the light through a prism.
The prism splits the white light into wavelengths of a different color. In a rainbow, the water droplets in the atmosphere act as a prism. Each individual wavelength within the spectrum of visible light wavelengths is representative of a particular color. When the light of that particular wavelength strikes the retina of our eye, we perceive that specific color sensation. When all of the colors strike our retina at the same time, we perceive white. Thus, the name white light.
This chart shows the colors within the visible light spectrum. Again, as with the entire electromagnetic spectrum, the shorter the wavelength, the greater the energy and temperature. The wavelengths in the visible light spectrum range in length from approximately 780 nanometers (7.80 x 10-7 m) down to 390 nanometers (3.90 x 10-7 m).
Sir Isaac Newton discovered that shining white light through a prism separated the wavelengths and displayed the true colors. This phenomenon is called dispersion.
You will see the visible light spectrum and the color associated with it. You may already know the phrase ROYGBIV to help remember the color and order of the waves in the visible light spectrum. (red, orange, yellow, green, blue, indigo, and violet).
Why Can’t We See UV Rays or Infrared Light?
We know that the light waves on the visible portion of the electromagnetic spectrum are detected by the rods and cones in our eyes. This stimulation causes an electric pulse to be sent to our brain, where our brain interprets the information as. “I am seeing red.” or “I am seeing blue.”
Why doesn’t our brain generate the message, “I’m seeing UV rays?” There are radio waves, ultraviolet rays, and infrared rays all around us. Why can’t we see them?
Simply put our rods and cones cannot detect the wavelengths of any of the other waves on the electromagnetic spectrum. Our eye receptors are not sized to receive the wavelength of gamma rays, ultraviolet light, infrared, or radio waves. Human eyes are the perfect size for only detecting and seeing the wavelengths in the visible or white light portion of the spectrum.
Scientists have developed infrared goggles to help the military carry out night operations. Night vision goggles have a lens that can turn infrared light into a wavelength that rods and cones can see and turn into an electrical message that can be sent to the brain for interpretation. These infrared sensing lenses are colored amber or green, so we can see infrared light. However, infrared light is not naturally amber or green.
If radio, infrared, and ultraviolet rays are all around us, why don’t we get burned?
If all of these waves of energy are bombarding the Earth from space, how can humans, animals, and plants continue to survive?
Approximately 29% of the solar energy reaching the top of the Earth’s atmosphere is reflected back to space by clouds, atmospheric particles, and reflective surfaces like sea ice and snow.
Water vapor, dust, ozone absorb about another 23% of the incoming solar energy.
About 48% of the solar energy passes through our atmosphere and is absorbed by the Earth’s surface (this is described in detail below.)
This means that approximately 71% of the incoming solar energy is somehow absorbed by the Earth and its systems.
The Earth’s atmosphere protects against certain types of electromagnetic radiation, including radio waves, infrared radiation, and some ultraviolet (UV) radiation. The extent of this protection varies depending on the specific wavelength of the radiation.
Radio waves: The Earth’s atmosphere is transparent to most radio waves, allowing them to pass through with minimal absorption or scattering. This is why we can receive radio signals from distant sources. The atmosphere does interact with extremely low-frequency (ELF) waves, but they are not harmful to human health.
Infrared radiation: The Earth’s atmosphere contains water vapor and carbon dioxide molecules that can absorb and re-emit infrared radiation. This absorption helps to prevent excessive heating of the Earth’s surface by trapping some of the infrared energy and contributing to the greenhouse effect. However, the atmosphere is not completely opaque to infrared radiation, and a significant amount of it can still reach the surface. This is why we feel the warmth from the Sun and other sources of infrared radiation.
Ultraviolet (UV) radiation: The Earth’s atmosphere provides protection against a substantial portion of the Sun’s UV radiation.
The atmosphere is the mixture of gases and other materials that surround the Earth in a thin, mostly transparent shell. It is held in place by the Earth’s gravity. The main components are nitrogen (78.09%), oxygen (20.95%), argon (0.93%), and carbon dioxide (0.03%). The atmosphere also contains small amounts, or traces, of water (in local concentrations ranging from 0% to 4%), solid particles, neon, helium, methane, krypton, hydrogen, xenon, and ozone.
The study of the atmosphere is called meteorology.
Life on Earth would not be possible without the atmosphere. Obviously, it provides the oxygen we need to breathe. But it also serves other important functions. It moderates the planet’s temperature, reducing the extremes that occur on airless worlds. For example, temperatures on the moon range from 120 °C (about 250 °F) during the day to -170 °C (about -275 °F) at night. The atmosphere also protects us by absorbing and scattering harmful radiation from the sun and space.
The ozone layer is located in the stratosphere, approximately 10 to 50 kilometers above the Earth’s surface, and plays a crucial role in absorbing UV-B and a portion of UV-C radiation. The ozone molecules absorb the high-energy UV radiation, preventing it from reaching the Earth’s surface. This protective layer shields us from the most harmful UV radiation, which can cause sunburn, skin damage, and an increased risk of skin cancer.
In addition to the ozone layer, other atmospheric components, such as water vapor and clouds, scatter and absorb some of the Sun’s radiation, further reducing the amount of energy reaching the Earth’s surface.
The Earth’s atmosphere also acts as an insulator, trapping some of the heat energy from the Sun and preventing it from escaping back into space. This natural greenhouse effect helps to maintain a relatively stable temperature range on Earth, making it suitable for life as we know it.
How does our atmosphere absorb electromagnetic energy?
The Earth’s atmosphere, you know that the stratosphere contains the ozone layer about 10-25 miles above the surface of the Earth.
The shape of an ozone molecule is such that it interferes with the waves in the ultraviolet band in the electromagnetic spectrum. Thus, the ozone layer is able to block some of the UV rays. Unfortunately, if you’ve ever suffered from sunburn, you know that some UV rays do get through the stratosphere and its ozone layer.
The second reason why we are protected from electromagnet energy is that the energy is not coming at us in a heavy bombardment. For example, if a friend had a toy gun that shot plastic balls, shooting one lightweight plastic ball at your leg wouldn’t hurt. However, if your friend fired off thousands of plastic balls at one time, you would feel pain in your leg!
The amount of electromagnetic radiation coming at us is NOT so intense as to cause harm. The Earth’s atmosphere, clouds, and the Earth’s surface have reflected about 30% of the electromagnetic radiation coming at the Earth. This is why X-ray and infrared telescopes are placed on high mountain tops or in orbit. Our atmosphere blocks out a large portion of these waves that telescopes cannot obtain optimal readings.
The Earth’s Albedo
The amount of radiation reflected back into space from the Earth’s surface and atmosphere is known as the Earth’s albedo. The Earth’s albedo varies depending on several factors, including the type of surface (land, ocean, ice), cloud cover, and atmospheric composition. On average, about 30% of the incoming solar radiation is reflected back into space.
Albedo is a measure of the reflectivity of a surface or object. It quantifies the percentage of incoming solar radiation that is reflected back into space. The term “albedo” is derived from the Latin word for “whiteness” and is often used in the context of the Earth’s energy balance.
Albedo is expressed as a value between 0 and 1, or as a percentage between 0% and 100%. A value of 0 means that the surface absorbs all of the incoming radiation, while a value of 1 (or 100%) indicates that all of the radiation is reflected.
Different surfaces and materials have different albedo values. Generally, surfaces that are lighter and more reflective have higher albedo values, while darker surfaces have lower albedo values. For example, fresh snow and ice have high albedo values because they reflect a significant amount of sunlight, whereas forests and oceans have lower albedo values because they absorb more radiation.
Albedo is an important factor in the Earth’s climate system. It affects the amount of solar energy absorbed or reflected by the Earth’s surface and atmosphere, influencing temperature patterns and climate. Changes in land surface conditions, such as melting ice or changes in vegetation cover, can alter the Earth’s albedo and have implications for climate change.
Here is a breakdown of the average albedo values for different surfaces and components:
- Land surfaces: Land surfaces have varying albedo values depending on factors such as vegetation cover, soil composition, and surface color. On average, land surfaces reflect about 10-30% of the incoming solar radiation.
- Ocean surfaces: The albedo of ocean surfaces depends on several factors, including the angle of sunlight, wind speed, and the presence of waves or currents. On average, oceans reflect about 6-20% of the incoming solar radiation.
- Snow and ice: Snow and ice have high albedo values, reflecting a significant portion of the incoming solar radiation. Fresh snow can reflect up to 80% of the radiation, while sea ice reflects about 30-50%.
- Clouds: Clouds play a crucial role in the Earth’s energy balance. They reflect a substantial amount of incoming solar radiation back into space. The albedo of clouds varies depending on their thickness, altitude, and composition. On average, clouds reflect about 30-80% of the incoming solar radiation.
UV Beads Experiment
UV beads are usually made of a type of plastic called polystyrene, which contains a special type of dye that is sensitive to ultraviolet light. The dye molecules in the uv beads change their shape and absorb UV radiation when exposed to sunlight or other sources of UV light, causing the uv beads to change color.
Where to Buy UV Beads
Over the years, I have purchased several different brands of UV beads. Two different brands are listed below.
Make a UV Beads Bracelet
Equipment
- UV beads At a minimum, I recommend giving each child 12 uv beads. The bead colors vary, so a minimum of 12 ensures they get at least one of each color.
- Chenille sticks
Instructions
- Give each student at least 12 beads.
- String the beads onto the chenille stick and close off the ends either as a bracelet or bent into another shape. (We've had students link chenille sticks and make necklaces.) Just let everyone have fun with this project!

- Test different ways to make the beads return to their "clear" state and then change colors. Place them in the sunlight, then in the shade. Place them under a jacket, or black construction paper, or a box. Go inside, then put them near a sunny window, near a shaded window. Put them outside for awhile then go into a darkened closet or room, do they glow in the dark. (Ours did.) Put sunscreen on them? Does this help? Test them over different days–when it's sunny, when it's cloudy, at night. What's also interesting is to test them on a sunny winter day vs. a sunny spring or summer day…at the same time of day. Take photos so you can compare the intensity of the colors in winter vs. summer.

In this video, the students covered up the uv beads with black construction paper and then white. When the uv beads were uncovered, they coloring changed quickly, so we have slowed down the video to show how much the uv beads did fade under the paper.
UV Beads Experiment – Light vs. Dark Covering
We tried several UV beads experiments. In one experiment the kids created, we covered our uv beads with black construction paper vs. white constructions paper.
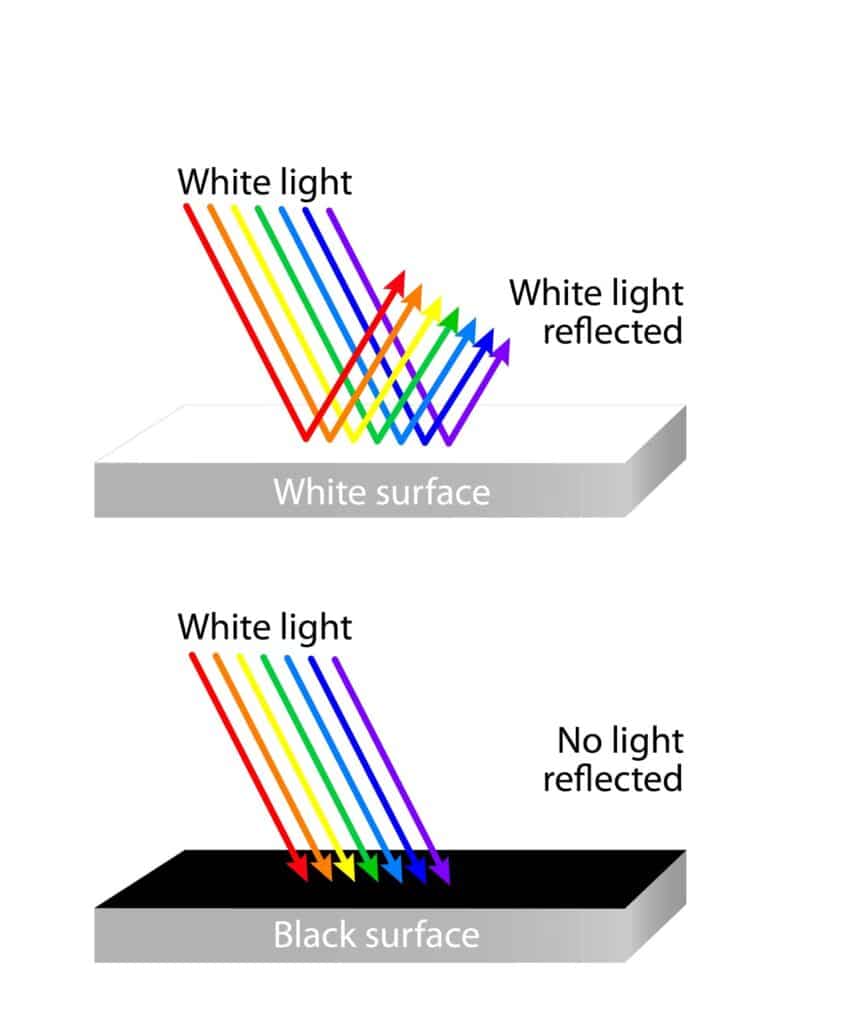
Since dark surfaces absorb more light than white surfaces, because they have a higher level of pigment or color, we hypothesized that when we covered the uv beads with the dark paper, the beads would get more pale than when they were covered with white paper.
When light hits a surface, it is either reflected, absorbed, or transmitted. Dark surfaces appear dark because they absorb most of the light that hits them, while white surfaces appear white because they reflect most of the light that hits them.
We know that light is made up of electromagnetic waves that have different wavelengths and energies. When light hits an object, the object’s color or pigment absorbs some of the wavelengths and reflects others. Dark surfaces have more pigment, which means they absorb more of the wavelengths of light that hit them, while white surfaces have less pigment, which means they reflect more of the wavelengths of light that hit them.
So, we were hoping to see a very obvious difference in the uv beads, even when we slowed down the video on the replay. Most of the students said they noticed that the uv beads were lighter when the black paper was lifted from the beads vs the white paper. We agreed we needed to do more testing.
We also discussed some factors that may have come that perhaps we need to control in another experiment like this. In addition to the color of the surface, the texture and angle of the surface can also affect how much light is absorbed or reflected. Rough surfaces tend to scatter light in different directions, while smooth surfaces tend to reflect light in a more predictable manner. The angle of the surface can also affect how much light is absorbed or reflected, as surfaces that are angled away from a light source will reflect less light than surfaces that are angled towards the light source.
Perhaps we held the papers at different angles? Could that have accounted for the difference.
There are a lot of different ideas and hypotheses to test in a uv beads experiment.
Additional Resources to Use with UV Beads Experiments
- NASA has online lessons, as well as some ebooks on the electromagnetic spectrum. These are definitely for grades 8-12.
- The Science of Light and Color for Kids video from Free School
I hold a master’s degree in child development and early education and am working on a post-baccalaureate in biology. I spent 15 years working for a biotechnology company developing IT systems in DNA testing laboratories across the US. I taught K4 in a private school, homeschooled my children, and have taught on the mission field in southern Asia. For 4 years, I served on our state’s FIRST Lego League tournament Board and served as the Judging Director. I own thehomeschoolscientist and also write a regular science column for Homeschooling Today Magazine. You’ll also find my writings on the CTCMath blog. Through this site, I have authored over 50 math and science resources.